This is taken from my book, Bread Science. I didn’t know how long it would take me to write up a new post, so I decided just to post this:
Sugar affects the rate of fermentation reactions. A little sugar, up to three percent, speeds up fermentation. The yeast processes the added sugar first, saving the time it would take to break down starch into sugar. With over three percent sugar, however, the fermentation rate no longer increases. [1] Above six percent, sugar actually decreases the rate. This is because the sugar begins to dehydrate the yeast cells. This effect, called crenation, was described in the “Salt and fermentation” section (copied below).
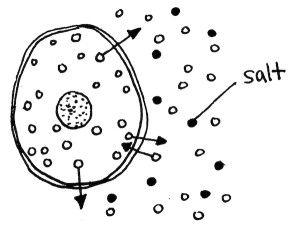
Salt slows fermentation reactions by dehydrating the yeast and bacteria cells. Without the nutrients they need, these cells cannot perform fermentation like usual.
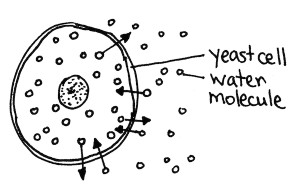
Basically, water molecules are able to pass in and out of cells, a process called osmosis (top left). When there is salty water outside of the cells, the salt interferes with the movement of the outside water molecules (top right). They pass into the cells more slowly. The inside water molecules are unaffected and pass out of the cell at their usual rate. Thus the net movement of water is out of the cells, an effect called crenation. This results in dehydrated yeast cells (bottom).
[1] Barham, H. N., Jr. and J. A. Johnson. “The influence of various sugars on dough and bread properties.” Cereal Chemistry 28 (1951) 463-473.

Pingback: Notes from “The Science of Yeast” Talk | Food Chem Blog
3% refers to what? Bakers percentage or sugar concentration? Of concern is how sweet can you make a liquid preferment. For example if I have 10oz. of flour and 15oz. of liquid in a preferment (sponge/poolish) , how much sugar or honey can I add that would not lead to yeast crenation in my starter?
Thanks.
Hi Neal,
I always thought the 3% referred to a baker’s percent (i.e., 3% sugar relative to 100% flour). I wanted to re-read the reference, though, to make sure, but I’m afraid I cannot find my copy at the moment, and I don’t have access online. What’s worrying me is that if the “players” in the crenation scenario are yeast, water, and sugar, it seems as if those relative amounts would be more important than the sugar:flour ratio. If the 3% IS baker’s percent, maybe it assumes a “normal” bread recipe (with about 60-70% water and 1% yeast).
Another thing to consider is that the yeast might eventually “catch up” in a sugary mixture. I do a demo in class with three containers with equal flour/yeast/water but differing amounts of sugar. I use lots of yeast to make it run quickly (and dramatically), so I can’t say it’s equivalent to a preferment with only a little yeast, but it still seems illustrative. The mixture with the most sugar starts very slowly, but eventually it begins to rise and then overflows just like the others. If your preferment is sitting overnight, having extra sugar might not be as much of an issue as it is in a dough, where you want it to rise in a few hours.
Now I’d like to mix some preferments with different percents and monitor them, to have data on the sugar percent that works best, as well as on if it makes a difference in a preferment. But in order to give you a timely reply, I will post this answer without data.
Perhaps mix in some very low concentration/high sweetness stevia if you’d like same texture/rise, but sweeter? If natural stevia volume is less than the partnered sugar volume, its potential bitterness is undetectable.
i know this is a litte late- but 35 of however much of yeast u are using
Thank you Ms. Beuhler. A week ago I somehow completely killed a sponge, not a single bubble. I had made it less hydrated by using some of the liquid for a soaker. I had suspected its demise was from an overly strong honey concentration, and a day or so later when I read that section in Bread Science, my suspicion was confirmed. Whether bakers percentage or solution concentration, I plan to reduce the honey in my sponges, which are now at 133% hydration, and usually ready to use in 4-6 hours.
Thanks again for your reply. This very topic is being discussed in the Perfect Sourdough group forum on Facebook. I hope you don’t mind that I have posted a link to this page.
Thanks Neal! I’m glad you figured out what was going on in your sponge.
Very helpful for my science project. Thanks
If I want sweeter bread can I double the amount of sugar and yeast?
Hi Karen, Sorry for my delayed reply. If the sugar gets over 6% (relative to the weight of the flour), it will slow the yeast activity, even if you add more yeast. All of the yeast will be affected. So you might see a slight increase in rising speed with doubled yeast, but the high sugar percentage will still control the dough’s rising rate. A better solution is to use “osmotolerant” yeast that is a strain of yeast that can withstand the higher sugar percentage. Or, if all you have is regular yeast, if you wait long enough, the dough will eventually begin to rise and rise enough; it just has a delay.
Will yeast fermentation still occur without sugar? Thanks!
Hi Connie, Even if you do not add sugar to bread dough, yeast fermentation will occur. The flour contains a tiny amount of sugar, but it also contains starch. The flour and yeast contain the enzymes needed to break down the starch into sugar. This sugar is then fermented. (If there is no sugar at all—for example, in a substance other than bread dough—I do not think fermentation can occur.) I hope this answers your question, and I am sorry for my delayed reply. Emily
Do you now know what the 3% refers to?
Hi Jasoooon, I’m afraid I have not put any time into finding an answer. I’m sorry to not be more helpful.
Emily
The 3% refers to flour ‘on weight’
Example….if you have 3kgs of flour, you will use the equation 3,000 (Flour weight) x 3% (the weight of sugar you need) = 90gms
Hello!
If the sugar is converted in alcohol and CO2 during the fermentation. How much of that 3% sugar is still remain in the dough after the fermentation?
Thanks
Hi Ana, I think it would be very hard to calculate this, because while the original sugar is being used up, new sugar is being produced (from the starches breaking down). When you bake the bread (and thus stop the yeast activity), it’s like you halt the process in the middle. But, even if you let the dough ferment until the yeast stops producing gas, there might still be sugar in the dough that was simply not near a yeast cell, and therefore not “eaten.” It’s such a complicated process!
How long will 2 quarts of 100 degrees water 2 1/2 cups of sugar and .25 oz of yeast Produce CO2? I use this Mixture in my diy bed bug traps And need to know how often I need to change this Mixture to keep Attracting bedbugs with the CO2.
Hi Tony, It’s been a while (decades!) since I calculated something like this. I’d have to dust off my general chemistry textbook and refresh my memory, and I’m afraid I don’t have the time for that. I think, though, that you don’t need someone with specialized bread knowledge to solve the problem. If I remember right, the calculation is done with the concentrations of the reactants, and the rate constant for the particular reaction, at the reaction temperature. (It’s tempting to try to figure it out, but I have deadlines to meet for work.) I hope you can find the answer you need.
Hi Emily, I am a beginner baker and few days ago, when I add more than 3% of sugar to my recipe, I noticed the fermentation rate has slowed down compared to some other recipe I made without adding sugar, and then I came across your post and finally realises too much sugar will slow down the rate of fermentation. Thank you for this post! Appreciated.
I’m glad the post helped!
Thanks for a wonderful post, Emily! How much of the sugar gets consumed by the yeast of you start with a 3% sugar sponge? I see nutrition information for bread recipes and they seem to assume all the starting sugar ends up as sugar in the final loaf. But wouldn’t the yeast convert it into complex carbs and protein? Thanks!
Hi Tim, You are right that yeast converts sugar (but into carbon dioxide and organic molecules like ethanol). When bread nutrition information lists sugar, I think it is because the amount is so much (like 1.5 grams of sugar per serving/slice), that the yeast does not process it all. I’m not exactly sure about your specific question, about a sponge with 3% sugar and how much would be consumed; that’s a pretty low amount, so that I think it might all be, but it would depend on the temperature (and thus rate of fermentation), and there might be sugar that is not consumed simply because it is not near a yeast cell. I’m feeling unsure of giving a definitive answer!
I’m trying to learn about baker’s percentage of sugar in bread dough and this post really helped me out! I’ve always wondered why some yeast brands have different types of yeast that cater to high-sugar and low-sugar recipes, and now I know! I do have a question though: so in the scenario where we use ‘normal’ yeast and add too much sugar, the fermentation is slowed down/delayed—but what happens if we apply the opposite to the yeast made for high sugar percentage? (Like, we use maybe 3-5% of sugar)
Hi Gian, Interesting question, I never considered this before! I think it should work fine: the high-sugar yeast is a strain that can continue functioning under the “tough” conditions of the higher osmotic pressure caused by the high sugar content, but should be able to function under the “easier” conditions without that osmotic pressure. But there might be something I’m not thinking of. If someone has tried this and found the high-sugar yeast to NOT function as well, please share.
Thank you for the explanation. I have a question…can you tell me how much yeast should be added for every addition of 4% sugar? Or do you have a formula for the optimal ratio of yeast to sugar in breadmaking?
Hi Julius, I’m sorry for my delayed reply. I think I understand that you are making a recipe with >6% sugar and do not have osmotolerant yeast, and you would like to add extra yeast to compensate. I’m afraid I don’t have any kind of formula for calculating how much. Another things to consider is that, while the reactions will be slower at first, eventually they will get going; you might end up with too much yeast activity or the yeast running out of food later in the process, if you add extra. I’ve not experimented to see how this situation might unfold; I’m basing this consideration on a demo I used to do at science fairs with three containers of dough with varying sugar levels. The one with a lot of sugar would barely react at first but eventually would start producing gas and rising. Sorry I cannot be more help with a definitive answer!
hi, can i ask what is the process of fermenting sugars using yeast
Hi Raven, I’m not sure I understand your question. If you’re looking for specifics of how a yeast cell breaks the sugar molecule into the products of fermentation, I’m afraid I can’t explain in more detail. I would have to re-read a biology textbook to find the details 🙂 If you’re asking about the process in terms of making bread, mixing the yeast into the dough gives it access to the sugars in the dough; the sugars are mostly from starch that breaks down into sugar.
Hi Emily! This really helped! Thanks!
I’m so glad to help!
Thank you so much Emily! This has allowed me to finally make good enough bread to feed the children!
No more starving kids hahahah
Lucky kids! I’m glad the post helped.
Emily you miracle worker! My sourdough starter has now become large enough to engulf the pot it was living in! Soon I hope it will gain sentience.
Many Thanks!
I’m glad your starter is thriving 🙂 Thanks for letting me know.
Can somebody tell me the limit of sugar wrt to flour weight for using low sugar or high sugar yeast ?
Hi Shreyasee, I believe the limit is 6% sugar relative to flour weight. Above this, adding sugar will slow the rate of fermentation if you don’t use osmotolerant (high sugar) yeast.